AGOMAB IS LEVERAGING NEW SCIENTIFIC INSIGHTS AND ESTABLISHED DRUG DEVELOPMENT EXPERTISE TO DEVELOP POTENTIAL DISEASE-MODIFYING THERAPIES ADDRESSING WELL-VALIDATED TARGETS AND HIGH UNMET NEED
We have built a focused pipeline of novel small molecule and antibody product candidates designed to act against well-validated and potentially disease-modifying targets for the treatment of fibrotic diseases with high unmet need and large commercial potential. We retain exclusive, worldwide development and commercialization rights to all of our product candidates and preclinical programs.
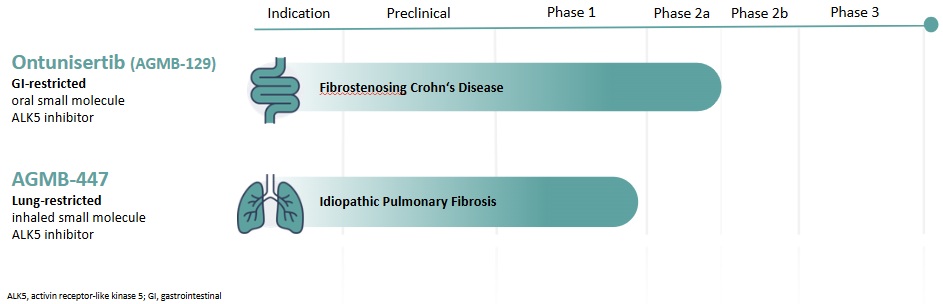
We have a robust discovery pipeline including several programs in the early stages of development. Our most advanced preclinical asset is AGMB-101.
“We are focused on advancing our pipeline of proprietary product candidates in fibrotic diseases with the goal of bringing truly disease-modifying treatments to patients in need.”
Philippe Wiesel, Chief Medical Officer

